A closed loop is a sealed circuit of water that circulates continuously without evaporation and is not exposed to the atmosphere.
Closed loops are commonly used to distribute chilled or heating water throughout buildings, or as condenser loops transferring heat between systems.
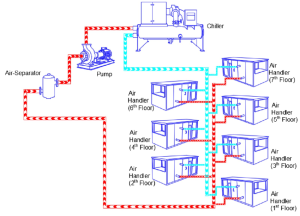
The two main risks in closed loops are corrosion and fouling.
If left untreated, the metals within the system will gradually corrode. The corrosion products formed will settle in low flow areas and on heat transfer surfaces, causing blockages, reduced heat transfer, increased pump load, and eventual equipment damage.
To prevent this, a closed-loop specific corrosion and scale inhibitor is added. This treatment adjusts the pH into the optimal range for the system metals and forms a protective film to prevent oxidation. Common inhibitors include sodium nitrite and azole compounds (such as tolyltriazole), which protect steel, copper, and other system metals.
These inhibitors are typically introduced into the system using a by-pass pot feeder, which allows safe chemical dosing without significant water loss.

Once dosed, the system must be periodically tested to ensure inhibitor levels remain within the required range.
Closed loops may require re-dosing for the following reasons:
- Water Losses
If a closed loop loses water, it is topped up with fresh mains water which contains no corrosion inhibitor.
This dilutes the system and reduces protection.
The system should be checked for leaks, repaired where necessary, then re-dosed to restore inhibitor concentration.
- Bacterial Activity (Nitrite-Reducing Bacteria)
Certain bacteria can convert sodium nitrite into sodium nitrate, which provides no corrosion protection.
This results in rapid corrosion if untreated.
If detected during routine testing, the loop must be dosed with a biocide to eliminate the bacteria, then re-dosed with corrosion inhibitor to restore protection.
